 The primary circuit of typical PWRs is divided into 4 independent loops (piping diameter ~ 700mm). The water (coolant) is heated in the reactor core to approximately 325☌ (⍴ ~ 654 kg/m 3) as the water flows through the core. The inlet temperature of the water is about 290☌ (⍴ ~ 720 kg/m 3). At this pressure, water boils at approximately 350☌ (662☏). Pressurized water reactors are cooled and moderated by high-pressure liquid water (e.g., 16MPa). Data do not represent any reactor design. It is an illustrative example, and the following data do not correspond to any reactor design. See also: Fluid Acceleration – Pressure Loss It differs by about 9% and therefore ice floats on liquid water It has a maximum density of 3.98 ☌ (1000 kg/m 3), whereas the density of ice is 917 kg/m 3. For example, water differs from most liquids in that it becomes less dense as it freezes. It must be noted and there are exceptions to this rule. Where ∆T is the temperature change, V is the original volume, ∆V is the volume change, and α V is the coefficient of volume expansion. The change in volume of a material that undergoes a temperature change is given by the following relation: This phenomenon is known as thermal expansion. However, the amount of expansion or contraction varies, depending on the material. Most substances expand when heated and contract when cooled. The effect of temperature on the densities of liquids and solids is also very important. 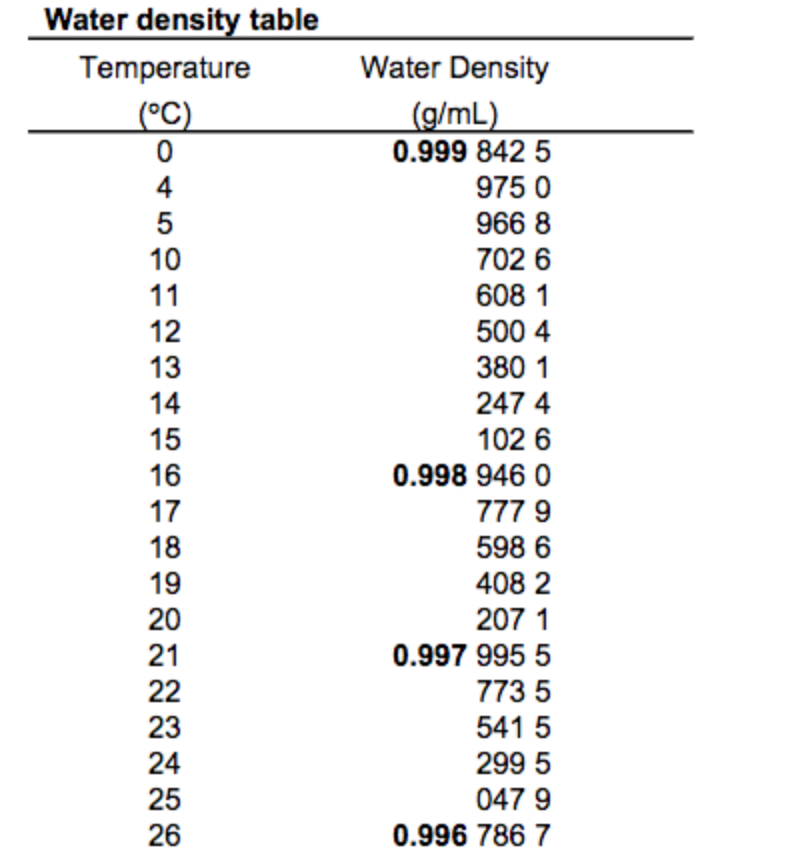
Compressibility measures the relative volume change of a fluid or solid as a response to a pressure change. On the other hand, the density of gases is strongly affected by pressure. The effect of pressure on the densities of liquids and solids is very small. Increasing the pressure always increases the density of a material. doi: 10.In general, density can be changed by changing either the pressure or the temperature. Wolf H, Bettin H, Gluschko A (2006) Water density measurement by a magnetic flotation apparatus. Tanaka M, Girard G, Davis R, Peuto A, Bignell N (2001) Recommended table for the density of water between 0 and 40 ☌ based on recent experimental reports. Masui R, Fujii K, Takenaka M (1995/96) Determination of the absolute density of water at 16 ☌ and 0,101325 MPa. Patterson JB, Morris EC (1994) Measurement of absolute water density, 1–40 ☌. Watanabe H (1991) Thermal dilatation of water between 0 and 44 ☌. Takenaka M, Masui R (1990) Measurement of the thermal expansion of pure water in the temperature range 0–85 ☌. Spieweck F, Bettin H (1992) Review: solid and liquid density determination. doi: 10.1021/je60064a005īettin H, Spieweck F (1990) Die Dichte des Wassers als Funktion der temperatur nach Einführung der internationalen temperaturskala von 1990. Kell GS (1975) Density, thermal expansivity, and compressibility of liquid water from 0 to 150 ☌: correlations and tables for atmospheric pressure and saturation reviewed and expressed on 1968 temperature scale. 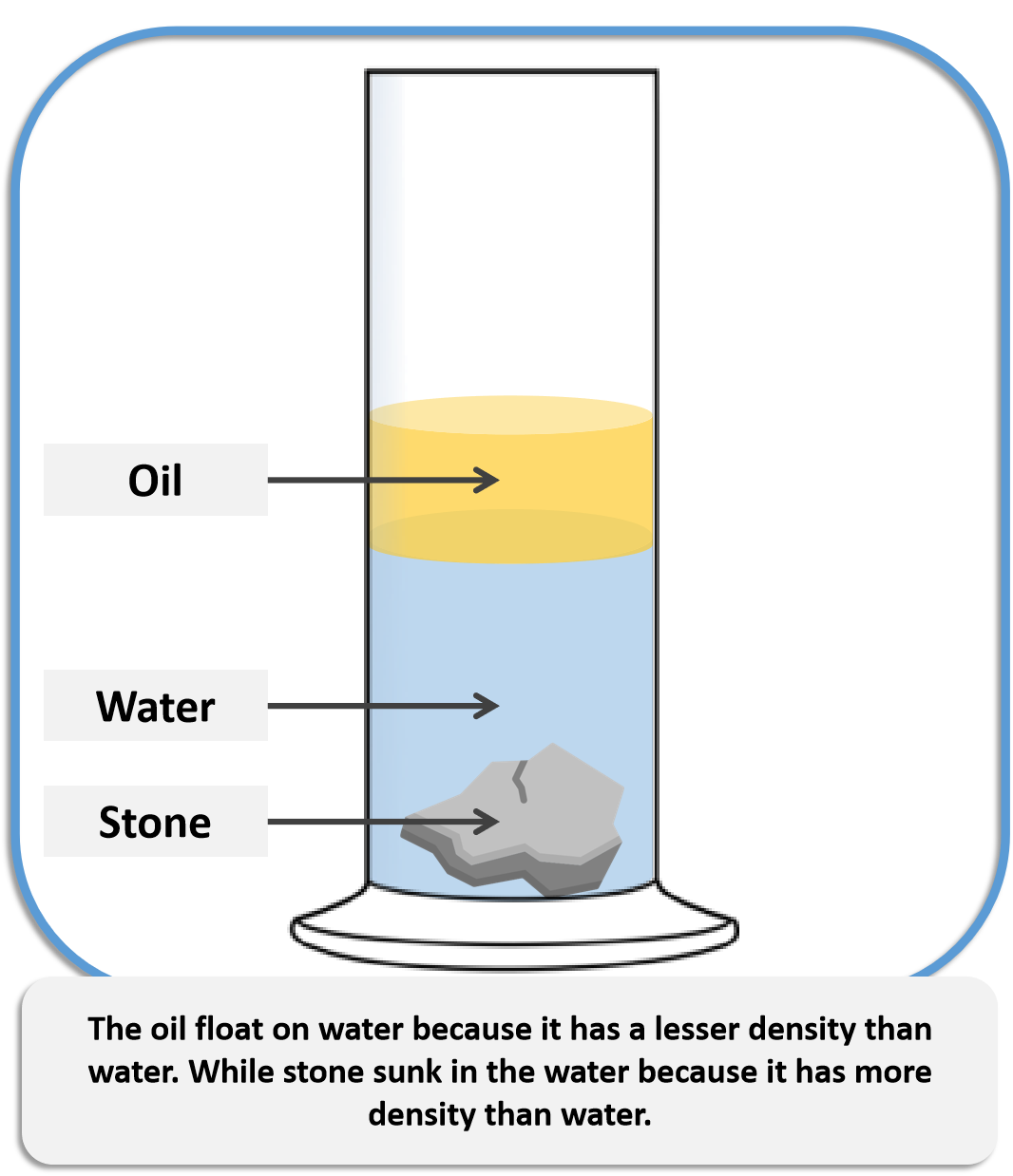
Wiss Abh Phys Tech Reichsanst 4:1–32Ĭhappuis P (1907) Dilatation de l’eau. Thiesen M (1918) Untersuchungen über die thermische Ausdehnung von festen und tropfbar flüssigen Körpern-bestimmung der Ausdehnung des Wassers für temperaturen zwischen 50° und 100°. Thiesen M, Scheel K, Diesselhorst H (1900) Untersuchungen über die thermische Ausdehnung von festen und tropfbar flüssigen Körpern-bestimmung der Ausdehnung des Wassers für die zwischen 0° und 40° liegenden temperaturen. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
